Metformin
Glucophage · Glumetza · Fortamet · Riomet · metformin HCl · 1 · 1-dimethylbiguanide
Last updated
At a glance
Overview
Metformin is the quietest workhorse in the physique-and-longevity toolkit. It's cheap, prescription-legal, and has more high-quality outcome data behind it than almost anything else you can stack — which is why it shows up in three very different contexts: on-cycle glucose management for guys running GH, heavy orals, or slin; as a recomp adjunct to sharpen insulin sensitivity during a cut; and as the backbone of most serious longevity stacks alongside rapamycin and low-dose tadalafil.
The mechanism is what makes it useful beyond diabetes. Metformin mildly inhibits mitochondrial complex I, raising the AMP:ATP ratio and activating AMPK — the master "low-energy" sensor that shuts down hepatic gluconeogenesis, suppresses lipogenesis, and improves GLUT4 translocation in skeletal muscle. Translation: lower fasting glucose, better partitioning of the carbs you eat, improved lipids, and a favorable shift in insulin sensitivity — all without driving hypoglycemia on its own.
"Intensive blood-glucose control with metformin substantially reduced the risk of diabetes-related endpoints, all-cause mortality, and myocardial infarction in overweight patients with type 2 diabetes." — UK Prospective Diabetes Study (UKPDS) Group, Lancet (1998)
There's one honest caveat worth flagging up front: if you're a natural lifter actively training for hypertrophy, the MASTERS trial showed chronic metformin can blunt the muscle-growth response to resistance training, likely via AMPK suppression of mTORC1. On a real cycle this signal disappears into the noise, but it's a real trade-off in a natural context and we'll come back to it. In this guide we'll cover the practical dose ladder (including why community doses are often lower than clinical ones), how to time it around meals and training, how it stacks with GH, insulin, GLP-1 agonists, and AAS, the side effects that actually matter (GI tolerance, B12, the lactic acidosis question), and how to decide whether metformin belongs in your protocol at all.
How Metformin works
Complex I Inhibition and the AMP:ATP Shift#
Metformin's upstream mechanism is a mild, reversible inhibition of mitochondrial respiratory chain Complex I in hepatocytes and, to a lesser extent, skeletal muscle. By throttling oxidative phosphorylation slightly, it drops cellular ATP and raises AMP — and that shifted AMP:ATP ratio is the signal that triggers almost everything downstream. This is why metformin is a "metabolic dimmer switch" rather than a hormone: it doesn't bind a receptor, it nudges the energy-sensor machinery.
AMPK Activation and Hepatic Gluconeogenesis#
The elevated AMP allosterically activates AMP-activated protein kinase (AMPK), the master energy-status kinase. Activated AMPK shuts down hepatic glucose output by suppressing gluconeogenic gene expression (PEPCK, G6Pase) via CRTC2, while simultaneously inhibiting lipogenesis through ACC phosphorylation. The practical read for physique users: fasting glucose drops, hepatic insulin resistance improves, and carb meals partition better — particularly useful when GH or heavy orals are pushing fasting glucose upward on bloodwork.
"Metformin was found to activate AMPK in rat hepatocytes, and this activation led to inhibition of gluconeogenic gene expression and glucose output." — Zhou G. et al., Journal of Clinical Investigation, 2001
AMPK-Independent Suppression of Gluconeogenesis#
Not all of metformin's glucose-lowering effect runs through AMPK. At clinically relevant concentrations it also inhibits mitochondrial glycerophosphate dehydrogenase (mGPD), breaking the glycerophosphate shuttle and blocking the conversion of lactate and glycerol into glucose. This is a parallel brake on gluconeogenesis that explains why metformin still works in AMPK-knockout models.
"Metformin inhibits mitochondrial glycerophosphate dehydrogenase, reducing the conversion of lactate and glycerol to glucose, thus revealing an AMPK-independent mechanism for the suppression of gluconeogenesis." — Madiraju A.K. et al., Nature, 2014
Skeletal Muscle Glucose Uptake and mTORC1 Cross-Talk#
In skeletal muscle, AMPK activation drives GLUT4 translocation to the sarcolemma, improving insulin-independent glucose uptake — the mechanism that makes metformin a credible glucose disposal agent around carb meals. The trade-off: AMPK also inhibits mTORC1, the primary hypertrophy signal. This is why chronic metformin can blunt the muscle-building response to resistance training in natural, untrained subjects — a real but manageable cost that mostly disappears in the presence of exogenous anabolics.
"The addition of metformin to PRT contributed to a smaller increase in lean mass and leg muscle hypertrophy compared to placebo." — Walton R.G. et al., Aging Cell, 2019
Gut-Level Effects: GLP-1, Microbiome, and Appetite#
A large fraction of metformin's action happens before it ever reaches the liver. In the gut it increases GLP-1 secretion, shifts the microbiome (notably expanding Akkermansia muciniphila), and modestly suppresses appetite. This is part of why lower "longevity" doses (500–1,000 mg/day) still produce meaningful metabolic effects, and why GI side effects — nausea, loose stools — are the most common complaint during titration. It's also the mechanism that pairs cleanly with GLP-1 agonists: the two hit glucose control from complementary angles without stacking hypoglycemia risk.
B12 Absorption and the Long-Term Caveat#
Chronic metformin use interferes with calcium-dependent B12 absorption in the terminal ileum. Over years this can drop serum B12 into frank deficiency, producing the neuropathy and fatigue that get misattributed to age or overtraining. The fix is cheap and preemptive: 500–1,000 mcg sublingual methylcobalamin daily, or an IM shot quarterly, plus an annual B12/MMA panel on anyone past the two-year mark.
"Long term use of metformin was associated with an increased risk of vitamin B-12 deficiency and lower serum concentrations of B-12." — de Jager J. et al., BMJ, 2010
Protocol
| Level | Dose | Frequency | Notes |
|---|---|---|---|
| Low | 500–1000 mg | Twice daily | Documented entry-level range |
| Mid | 1000–1500 mg | Twice daily | Most commonly studied range |
| High | 1500–2000 mg | Twice daily | Start 500 mg once daily with the largest meal; titrate by 500 mg/week to target. XR can be dosed once daily; IR is typically split AM/PM with meals. Longevity users often stay at 500–1,000 mg/day. |
Cycle length & outcomes
Documented cycle
4–52 weeks
Plateau after
24 wks
Cycle Structure & Duration#
Metformin is not a traditional "cycle" compound — there's no HPTA suppression, no receptor downregulation, no PCT. It's run continuously for as long as it's serving a purpose, with GI tolerance dictating the on-ramp and bloodwork dictating the off-ramp (if any). The only mandatory tapering is up, not down: start low to avoid a week of diarrhea.
| Goal | Cycle Length | Daily Dose | Timing |
|---|---|---|---|
| On-cycle glucose disposal (AAS ± GH) | Entire blast (8–20 weeks) | 500–1,000 mg | XR w/ dinner, or 500 mg BID |
| GH + slin stack (advanced) | Duration of GH/slin use | 1,000–1,500 mg | 500 mg with each major carb meal |
| Cutting / recomp adjunct | Final 6–10 weeks of cut | 1,000–1,500 mg | Split lunch + dinner |
| PCOS / androgen-driven acne (female) | 12+ weeks, ongoing | 1,500–2,000 mg | Split AM/PM with meals |
| Longevity / healthspan | Indefinite (years) | 500–1,000 mg | XR w/ dinner |
| Pre-workout GDA only | Session-based | 500–1,000 mg | With pre-workout meal |
Titration Schedule#
The single most important protocol detail is the ramp. Jumping straight to 2,000 mg is how people decide metformin "isn't for them" after 48 hours on the toilet.
| Week | Dose | Notes |
|---|---|---|
| 1 | 500 mg once daily | With largest meal (usually dinner) |
| 2 | 500 mg BID or 1,000 mg XR | Add breakfast dose, or switch to XR once daily |
| 3 | 1,500 mg/day | If targeting higher range |
| 4+ | 2,000 mg/day | Therapeutic ceiling for most users |
XR vs IR: Default to XR unless cost forces otherwise. GI tolerance is dramatically better, once-daily dosing is cleaner, and the pharmacokinetics are smoother across the day. IR is fine but unnecessarily rough for most people.
Onset Timing#
- Fasting glucose / insulin: measurable shift within 3–5 days, stable by week 2.
- HbA1c: meaningful drop by 8–12 weeks (HbA1c reflects ~90-day glucose average, so earlier measurement is noise).
- Body composition effects: subtle. The 2–3 kg weight reduction seen in clinical trials takes 12+ weeks to materialize and is mostly appetite-mediated.
- GI side effects: peak in the first 1–2 weeks, resolve in >80% of users by week 3 if titrated properly.
"Metformin was found to activate AMPK in rat hepatocytes, and this activation led to inhibition of gluconeogenic gene expression and glucose output." — Zhou et al., J Clin Invest 2001
Bloodwork Cadence#
Metformin is one of the easier compounds to monitor because renal function and B12 are the only two numbers that really matter long-term.
| Marker | Frequency | Why |
|---|---|---|
| Fasting glucose + HbA1c | Every 3–6 months on cycle | Track partitioning and on-cycle IR |
| Fasting insulin / HOMA-IR | Every 6 months | Confirms mechanism is working |
| Serum creatinine / eGFR | Yearly (every 6 months if >50yo) | Hard stop at eGFR <30 |
| Serum B12 + MMA | Yearly after year 2 | Ileal absorption drops over time |
| Lipid panel | Every 6 months on cycle | Baseline improves; useful alongside AAS-driven shifts |
"Long term use of metformin was associated with an increased risk of vitamin B-12 deficiency and lower serum concentrations of B-12." — de Jager et al., BMJ 2010
Supplement 500–1,000 mcg methylcobalamin sublingual daily as cheap insurance from month six onward. It costs nothing and removes the one predictable chronic side effect from the equation.
When to Pause or Stop#
- 48 hours around IV iodinated contrast (CT with contrast, angiography). Standard hospital practice.
- Acute GI illness with dehydration — the only realistic lactic acidosis scenario. Hold until hydrated and eating normally.
- Heavy drinking episodes — skip the dose. Moderate drinking alongside metformin is fine; binges are the risk factor.
- eGFR drops below 30 — stop. Between 30–45, reduce to 500–1,000 mg/day and monitor.
No taper required to stop. Hepatic gluconeogenesis resumes normal output within 24–48 hours of discontinuation.
The MASTERS Caveat (Natural Lifters)#
One honest trade-off worth stating plainly:
"The addition of metformin to PRT contributed to a smaller increase in lean mass and leg muscle hypertrophy compared to placebo." — Walton et al., Aging Cell 2019
If you're natural, untrained, and the primary goal is hypertrophy — metformin's AMPK-driven mTORC1 suppression is working against you. Consider berberine (weaker AMPK activation, no prescription) or just skip glucose-disposal agents entirely. On a real AAS cycle this signal is drowned out by exogenous anabolic drive and the on-cycle glucose-control benefit wins.
Indefinite Use#
Unlike almost everything else on this site, metformin has a 25+ year safety record at therapeutic doses, outcome data from UKPDS 34 (Lancet 1998) showing reduced all-cause mortality in overweight T2D patients, and no evidence of tolerance or escape. Users running it for longevity purposes stay on 500–1,000 mg/day for years without issue, pausing only for contrast imaging or acute illness. The ceiling on duration is renal function, not the drug itself.
Body Transformation Preview
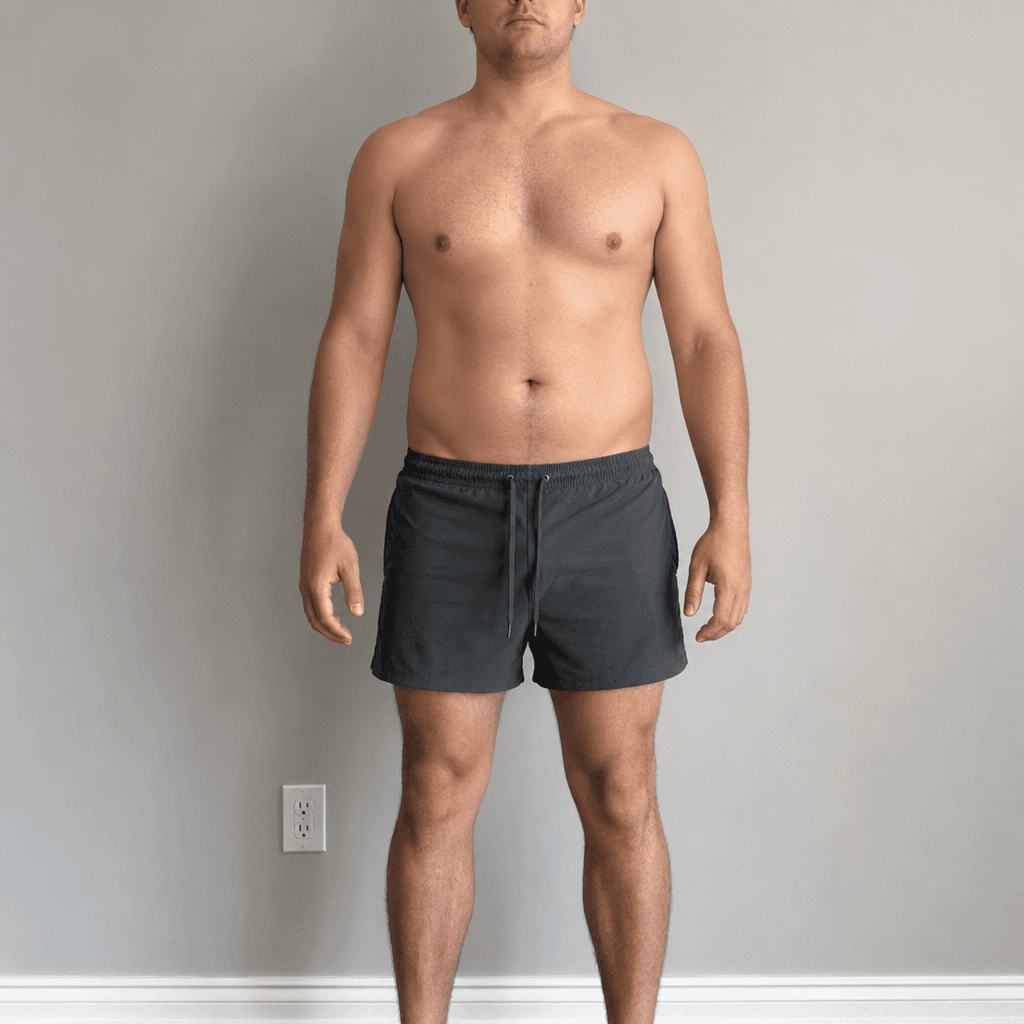
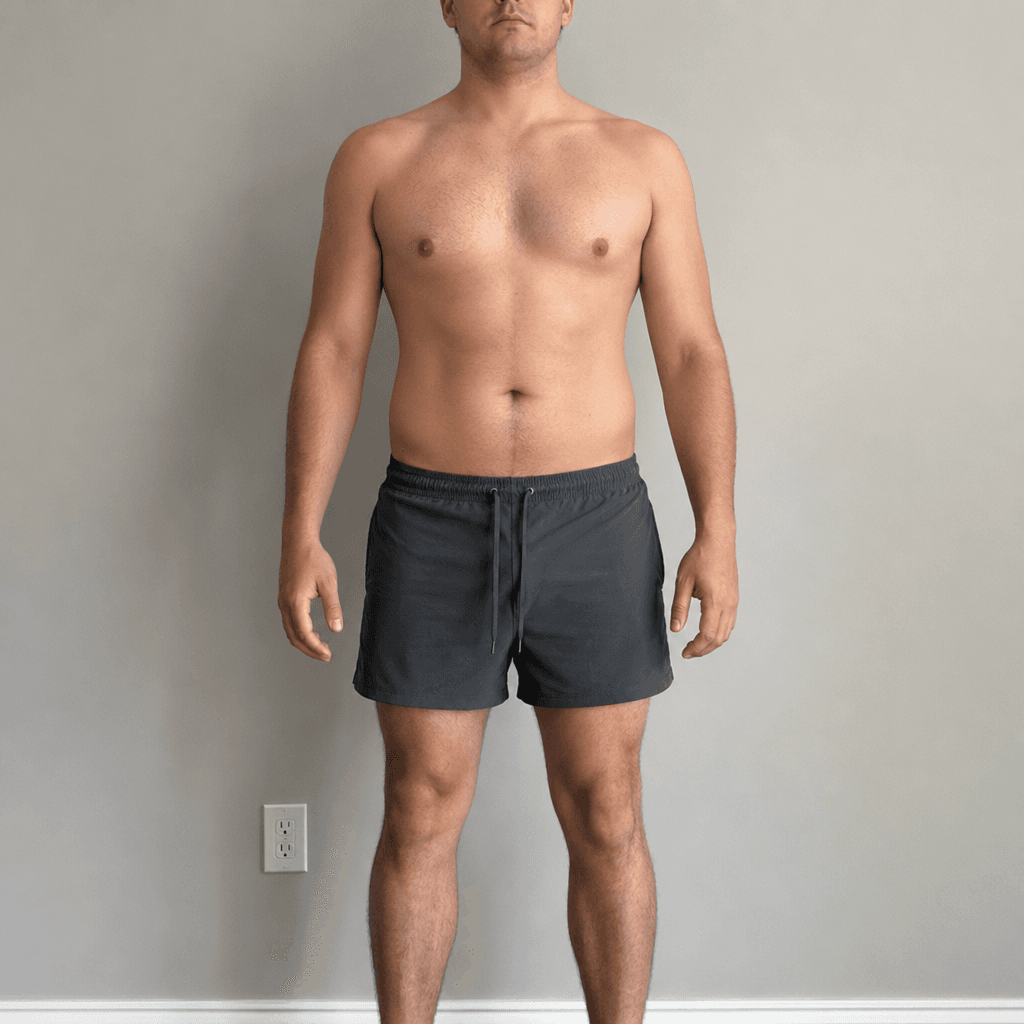
Lean Mass Gain
0.0 lbs
0.0–0.0 lbs range
Fat Loss
5.8 lbs
4.3–7.2 lbs range
Fat Loss by Week
Risks & mistakes
Common (most users)#
- GI distress — nausea, diarrhea, cramping, flatulence, metallic taste. Hits roughly 20–30% of starters and is almost entirely a titration problem. Start at 500 mg once daily with the largest meal of the day and add 500 mg per week until you hit target dose. Administering the dose mid-meal, rather than pre-meal, matters. Switch from IR to XR (extended-release) if symptoms persist — this alone resolves it for most people.
- Appetite suppression. Mild, mostly welcome on a cut. Not a reason to dose-adjust unless you're in a bulk and struggling to hit calories, in which case split the dose across more meals or drop to 500 mg/day.
- Soft stools / looser bathroom schedule even after the initial GI phase resolves. Usually dose-dependent; dropping from 2,000 to 1,500 mg/day fixes it.
- Mild fatigue or reduced aerobic ceiling in the first few weeks. Typically subsides; if it doesn't, see the Konopka data below.
Uncommon (dose-dependent or individual)#
- Vitamin B12 deficiency. Real, documented, and slow-moving — shows up on long-term use (>2 years, especially >1,500 mg/day). Mechanism is reduced ileal B12 absorption. Supplement 500–1,000 mcg methylcobalamin sublingual daily, or 1 mg IM quarterly, on any run longer than a year. Check serum B12 and methylmalonic acid yearly. Watch for paresthesias, glossitis, or unexplained fatigue.
"Long term use of metformin was associated with an increased risk of vitamin B-12 deficiency and lower serum concentrations of B-12." — de Jager et al., BMJ 2010
- Blunted aerobic training adaptations. Metformin modestly attenuates mitochondrial biogenesis in response to endurance work. If your goal is a VO2max PR or a marathon block, metformin is working against you. Pull it for the training cycle.
- Blunted hypertrophic response in a natural context. The MASTERS trial is the elephant in the room for natural lifters. On a real cycle this signal drowns out; if you're natural and actively trying to grow, it's a genuine trade-off.
"The addition of metformin to PRT contributed to a smaller increase in lean mass and leg muscle hypertrophy compared to placebo." — Walton et al., Aging Cell 2019
- Elevated lactate at rest. Usually subclinical and irrelevant, but if you're doing lactate-threshold testing or metabolic work your numbers will shift.
- Bloodwork to watch: serum creatinine and eGFR every 6–12 months (dosing ceiling falls as renal function does), B12 + MMA yearly, fasting glucose and HbA1c every 3–6 months on cycle to confirm it's actually doing its job.
Rare but serious#
- Lactic acidosis. The headline risk and the one people panic about. Actual incidence is <10 per 100,000 patient-years, and essentially every case occurs in someone with significant renal impairment, acute illness, severe dehydration, heavy alcohol binges, or a perfect storm of all four. Warning signs: deep labored breathing, severe muscle pain, unexplained nausea and weakness, hypothermia. Stop immediately and go to the ER. In a healthy physique user with normal kidneys this is a theoretical risk, not a practical one.
- Acute kidney injury from unrelated causes (rhabdo, severe dehydration, contrast, sepsis) will concentrate metformin and convert a theoretical lactic acidosis risk into a real one. Hold the drug during any acute illness involving vomiting, diarrhea, or low fluid intake.
Hard contraindications#
- eGFR <30 mL/min/1.73 m². Do not use. eGFR 30–45 requires dose reduction.
- Active heavy alcohol use / binge drinking. Lactic acidosis risk goes from theoretical to real. Moderate drinking is fine; weekend binges plus metformin is not.
- Severe hepatic impairment. Liver clears the lactate metformin generates; impaired liver + metformin = acidosis risk.
- Acute decompensated heart failure, sepsis, shock, or any state of tissue hypoperfusion.
- IV iodinated contrast imaging. Hold metformin 48 h before and after in anyone with any degree of CKD.
- Major surgery / prolonged NPO status. Hold for the perioperative window.
- Insulin + fasted training or skipped meals. This is not a metformin contraindication per se, but it's worth stating plainly: if you're running exogenous insulin, metformin does not protect you from hypoglycemia. Glucose tabs and real food do. Do not treat metformin as a safety net for sloppy slin protocols.
Gender-specific and PCT considerations#
Metformin has no hormonal activity, no HPG axis suppression, no virilization risk, and no PCT requirement — it's endocrine-neutral in both sexes. It is extensively used in women for PCOS at 1,500–2,000 mg/day and has a long track record of safe use in pregnancy (historically category B; not a teratogen in available data). No dose adjustment by sex. One of the cleanest metabolic tools in the toolkit — the side-effect profile is almost entirely GI, B12, and the renal/contrast housekeeping above.
Stack & combine
Multipliers applied when these compounds run together. Values > 1 indicate a bonus on that axis. Tap a partner to expand the mechanism.
| Partner | Type | Lean | Fat loss | Recovery |
|---|---|---|---|---|
| synergistic | ×1.05 | ×1.20 | ×1.12 | |
| additive | ×1.00 | ×1.08 | ×1.00 | |
| additive | ×1.00 | ×1.05 | ×1.00 |
Featured in stacks1 curated protocol include Metformin
FAQ — Metformin
Research & citations
5 studies cited on this page.
Conclusion
Metformin is a veteran mainstay for physique-focused users chasing better glucose control, insulin sensitivity, and metabolic health — with real evidence for longevity and on-cycle risk reduction if you leverage it right.
Key takeaways:
- Standard dose is 500–2,000 mg/day, titrated slowly; 1,000 mg/day (XR) with dinner is the community sweet spot for most
- XR (extended-release) is favored for far better GI tolerance
- Split dosing with meals (AM/PM) improves tolerability and steadier effect
- 4–52+ week run length — most users run continuously or in cut/cycle-specific blocks
- Pairs well with GH, insulin, orat GLP-1s for improved glucose disposal; minimal solo effect on fat loss (~0.15 lb/wk)
- Stack: add B12 (500–1,000 µg/d) on long-term use to offset deficiency risk (de Jager et al., BMJ 2010)
- Heads up for naturals: chronic use can blunt hypertrophy/mTOR signaling — especially notable off-cycle (Walton et al., Aging Cell 2019)
If your goal is to keep blood glucose in range, offset GH or oral-induced insulin resistance, or add a low-cost healthspan angle, metformin remains a staple — just respect its limitations and ramp up slow for GI comfort.