Oleoylethanolamide
OEA · oleoyl ethanolamide · N-oleoylethanolamine · NAE 18:1
Last updated
At a glance
Overview
Why OEA Earned Its Spot in the Cutting Toolkit#
Oleoylethanolamide is the endogenous lipid your small intestine already makes every time dietary fat hits the duodenum — a high-affinity PPAR-α agonist that tells the brain the meal is over and tells the liver to start oxidizing fat. Supplementing it exogenously turns that signal up on demand, which is exactly why physique-focused users reach for it during a cut, as a non-hormonal, non-stimulant lever on appetite and "food noise."
What makes OEA interesting to the bodybuilding and looksmaxxing crowd isn't a single dramatic effect — it's the combination. Published RCTs at 125 mg/day for 8–12 weeks show meaningful reductions in body weight, waist circumference, liver enzymes, fasting glucose, HOMA-IR, and inflammatory markers, alongside upregulation of the fat-oxidation machinery (UCP1, UCP2, PPAR-α itself). That makes it a useful adjunct whether the goal is polishing a cut, softening the rebound hunger at the tail of a GLP-1 week, or supporting liver numbers during a harsh oral cycle.
"Supplementation with OEA significantly decreased body weight, BMI, waist circumference, FBS, HOMA-IR, ALT, AST, GGT, TG, hs-CRP, TNF-α, and MDA, and increased the mRNA expression of PPAR-α, UCP1, and UCP2 compared to placebo." — Tutunchi et al., Pharmacological Research (2020)
Expect modest, cumulative effects rather than GLP-1-grade hunger annihilation — OEA shrinks meal size and stretches the inter-meal interval, it doesn't abolish appetite. The sections below cover the documented dosage ladder (125 mg clinical vs. 200–600 mg community), pre-meal timing, stacking logic with GLP-1 agonists and liver-support compounds, the side-effect profile, and the most common protocol mistakes — starting with dosing powder without any dietary fat and expecting anything to happen.
How Oleoylethanolamide works
PPAR-α Agonism — The Master Switch#
OEA is a high-affinity endogenous agonist of peroxisome proliferator-activated receptor-alpha (PPAR-α), a nuclear receptor concentrated in the small intestine, liver, and adipose tissue. PPAR-α engagement switches on the transcriptional program for fatty-acid β-oxidation, upregulates UCP1 and UCP2 (uncoupling proteins that dissipate energy as heat), and drives lipolysis in fat depots. This is the mechanism responsible for the weight, waistline, and liver-enzyme reductions seen in the clinical trial data.
"Supplementation with OEA significantly decreased body weight, BMI, waist circumference, FBS, HOMA-IR, ALT, AST, GGT, TG, hs-CRP, TNF-α, and MDA, and increased the mRNA expression of PPAR-α, UCP1, and UCP2 compared to placebo." — Tutunchi et al., Pharmacological Research, 2020
Practical translation: chronic dosing biases the metabolic machinery toward burning stored fat as substrate, which is why the compound earns its place in a cutting stack rather than a performance stack.
Peripheral Satiety — The Gut-Brain Relay#
Dietary fat in the proximal small intestine triggers enterocytes to synthesize OEA locally. That OEA then engages PPAR-α on vagal afferent nerves, which relays a satiety signal to the brainstem (NTS) and hypothalamic feeding circuits. The practical output: delayed meal initiation, smaller meal size, longer inter-meal intervals — the opposite of the food-noise phenotype that derails most cuts.
"OEA, an endogenous lipid, acts as a high-affinity agonist of PPAR-alpha and induces a remarkable reduction of food intake and body weight gain in obese rodents." — Fu et al., Neuropharmacology, 2005
Exogenous oral OEA piggybacks on this same pathway, which is why pre-meal administration with a small fat source outperforms fasted dosing — the dietary fat both solubilizes the compound and primes the gut-brain circuit OEA is designed to exploit.
GPR119 and Endogenous GLP-1 Potentiation#
Alongside PPAR-α, OEA activates GPR119 on enteroendocrine L-cells, which potentiates release of endogenous GLP-1 and PYY. This is a second, faster satiety arm that operates on meal-to-meal timescales rather than the slower transcriptional PPAR-α arm. It also explains why OEA stacks synergistically with exogenous GLP-1 agonists (semaglutide, tirzepatide, retatrutide) in rodent models — the two mechanisms converge on appetite but arrive via independent upstream receptors, so additivity is real rather than redundant.
For users running a low-dose GLP-1 protocol, OEA is a logical co-administered tool: it preserves endogenous incretin tone and helps blunt the rebound hunger that shows up toward the end of the weekly GLP-1 dosing window.
Fat Substrate Utilization#
Beyond reducing intake, OEA reshapes how the body handles the calories it does receive. PPAR-α activation upregulates carnitine palmitoyltransferase-1 (CPT-1) and the full β-oxidation enzyme cascade, shifting substrate preference toward fatty-acid oxidation. UCP1/UCP2 upregulation simultaneously increases non-shivering thermogenesis in brown and beige adipose.
"OEA administration reduces meal frequency and overall caloric intake while promoting utilization of stored fat as an energy source." — Brown et al., Physiology & Behavior, 2017
The two effects compound: lower intake plus higher oxidation of stored lipid. This is also the mechanism behind OEA's liver-enzyme improvements in NAFLD populations — hepatic steatosis regresses when the liver is transcriptionally pushed to oxidize rather than store fat, which is directly relevant for anyone coming off a heavy oral AAS block with elevated ALT/AST.
Central Monoamine Modulation#
OEA is not purely a peripheral compound. PPAR-α engagement in the CNS produces measurable increases in acetylcholine and serotonin (5-HT) in cortical regions, which plausibly underlies the mild wakefulness, mood-lift, and reduced hedonic food-seeking that users report anecdotally.
"PPARα engagement by OEA significantly increased both acetylcholine and serotonin (5-HT) extracellular levels in the cortex, supporting its neurochemical effects." — Murillo-Rodríguez et al., European Journal of Neuroscience, 2021
This neurochemical signature is why OEA is often deployed specifically against evening "food noise" and hedonic snacking — the compound dampens the reward-driven arm of eating alongside the homeostatic arm, rather than only suppressing raw hunger.
Rapid Hydrolysis, Durable Downstream Effect#
OEA is inactivated within ~30–60 minutes by fatty-acid amide hydrolase (FAAH) and N-acylethanolamine acid amidase (NAAA), which cleave it into oleic acid and ethanolamine. On paper this looks like a pharmacological weakness — the ligand is gone before the next meal. In practice, PPAR-α is a nuclear transcription factor: once activated, it alters gene expression (UCP1, UCP2, CPT-1, β-oxidation enzymes) on a timescale of hours to days. This is why a single daily pre-meal dose produces measurable metabolic remodeling over 8–12 weeks despite a sub-hour plasma half-life.
"Daily supplementation with 125 mg OEA led to improvements in glycemic indices, oxidative stress biomarkers, and serum inflammatory factors after 8 weeks with no adverse events reported." — Darvishi et al., Journal of Ovarian Research, 2024
The ligand is transient; the transcriptional rewiring is not. That asymmetry is what makes OEA a viable oral compound despite the rapid FAAH/NAAA clearance.
Protocol
| Level | Dose | Frequency | Notes |
|---|---|---|---|
| Low | 125–200 mg | Once daily | Documented entry-level range |
| Mid | 200–300 mg | Once daily | Most commonly studied range |
| High | 300–600 mg | Once daily | Administered 30–60 min before the largest meal of the day. Higher community protocols (400–600 mg) split across 2–3 pre-meal doses. Published RCTs use 125 mg single daily dose for 8–12 weeks. |
Cycle length & outcomes
Documented cycle
8–16 weeks
Plateau after
12 wks
Cycle Structure#
OEA is a non-hormonal metabolic adjunct — no suppression, no taper, no PCT. Cycle length is dictated by the diet phase it supports, not by receptor desensitization or endocrine recovery. The compound is run in 8–16 week blocks aligned to a cut, then dropped when the deficit ends or the goal weight is hit.
| Goal | Cycle Length | Daily Dose | Timing |
|---|---|---|---|
| Pre-meal appetite control (cut) | 8–12 weeks | 200–300 mg | 30–60 min before largest meal |
| Aggressive cutting / "food noise" damper | 10–16 weeks | 400–600 mg (split 2–3×) | Pre-meal, staggered |
| GLP-1 adjunct (semaglutide/tirzepatide stack) | matched to GLP-1 titration | 125–250 mg | Pre-breakfast |
| NAFLD / post-oral-AAS liver support | 12 weeks | 125 mg | With main meal |
| Recomp / clean-bulk appetite smoothing | 8–12 weeks | 125–200 mg | Pre-dinner |
| PCOS metabolic / inflammation (per RCT) | 8 weeks | 125 mg | With main meal |
Onset & Timeline#
OEA works on two clocks. The meal-suppressant effect is immediate — within the first dose, subjects report delayed onset of hunger and smaller portion size at the target meal. This is the acute PPAR-α → vagal afferent signal. The metabolic and inflammatory changes (ALT/AST, hs-CRP, TNF-α, HOMA-IR, lipids) are transcriptional and take weeks to manifest.
Published clinical readouts:
"Supplementation with OEA significantly decreased body weight, BMI, waist circumference, FBS, HOMA-IR, ALT, AST, GGT, TG, hs-CRP, TNF-α, and MDA, and increased the mRNA expression of PPAR-α, UCP1, and UCP2 compared to placebo." — Tutunchi et al., Pharmacological Research (2020)
"Daily supplementation with 125 mg OEA led to improvements in glycemic indices, oxidative stress biomarkers, and serum inflammatory factors after 8 weeks with no adverse events reported." — Darvishi et al., Journal of Ovarian Research (2024)
Expect:
- Week 1: reduced meal size, longer inter-meal interval, occasional mild nausea on higher community doses
- Weeks 2–4: stabilized appetite curve, measurable scale movement on a calorie deficit
- Weeks 6–8: liver enzyme and inflammatory marker improvements visible on bloodwork
- Weeks 8–12: peak PPAR-α / UCP1 / UCP2 transcriptional effects; this is where the NAFLD and metabolic numbers move hardest
Loading, Tapering & Frequency#
No loading phase. Unlike compounds that benefit from saturation kinetics (creatine, some peptides), OEA's effect is driven by the acute PPAR-α ligand concentration at mealtime plus downstream gene expression — front-loading produces no additional benefit and wastes compound.
No taper. OEA has a ~30–60 minute plasma half-life (rapid FAAH/NAAA hydrolysis), no hormonal suppression, and no documented withdrawal or rebound-hyperphagia pattern. The cycle simply ends.
Frequency: once-daily at the 125–300 mg range is fine. Above ~300 mg/day the community splits dosing across 2–3 pre-meal windows — this tracks the kinetics better, since a single oral dose clears before the next meal.
With fat. Unmicronized bulk powder is poorly soluble. A fish-oil capsule or ~5 g of dietary fat 30–60 min pre-dose materially improves absorption. The 125 mg clinical trial dose uses an encapsulated/formulated product; bulk-powder users typically need 2–3× that amount to match the subjective effect, which is why community protocols cluster at 200–300 mg.
Bloodwork Cadence#
Minimal. OEA is one of the cleanest metabolic adjuncts on the shelf.
- Baseline (before initiation): CMP (ALT, AST, GGT, glucose), fasting lipid panel, hs-CRP. This also serves as the reference for judging whether the compound is actually moving numbers.
- Week 8–12: repeat CMP + lipids + hs-CRP. This is where the trial data shows the strongest signal.
- On-cycle with oral AAS: fold OEA bloodwork into the existing mid-cycle liver panel — no separate schedule needed.
No androgen panel, no CBC monitoring, no renal-specific tracking required. Clinical trials at 125 mg/day for 8–12 weeks reported no significant adverse events versus placebo.
Stopping & Re-Running#
The compound can be cycled on and off indefinitely. There is no documented tolerance pattern — the 8-week and 12-week RCTs show sustained effect through the trial endpoint, and community users report consistent response across repeated cutting blocks. A common pattern is 12 weeks on during a cut, off during maintenance or bulk, back on for the next cut. No washout period is mechanistically required.
Body Transformation Preview
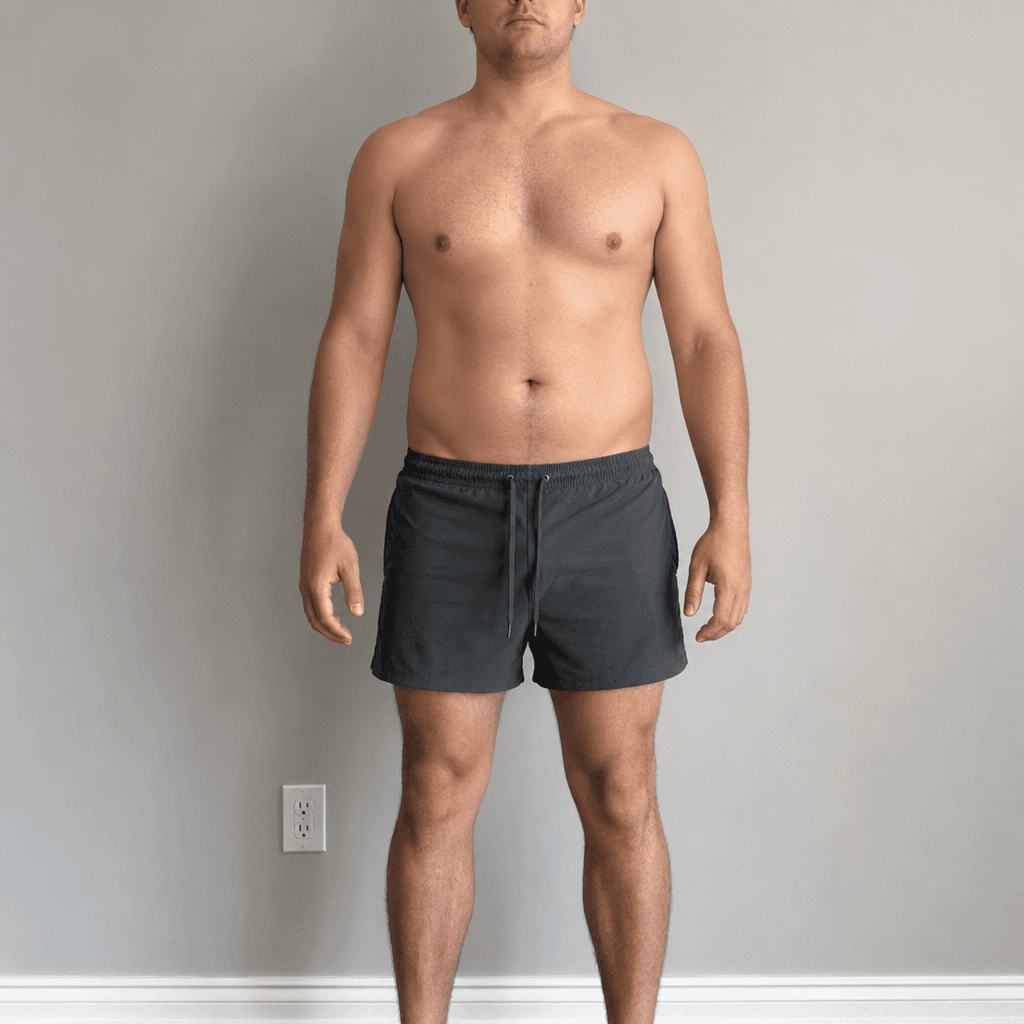
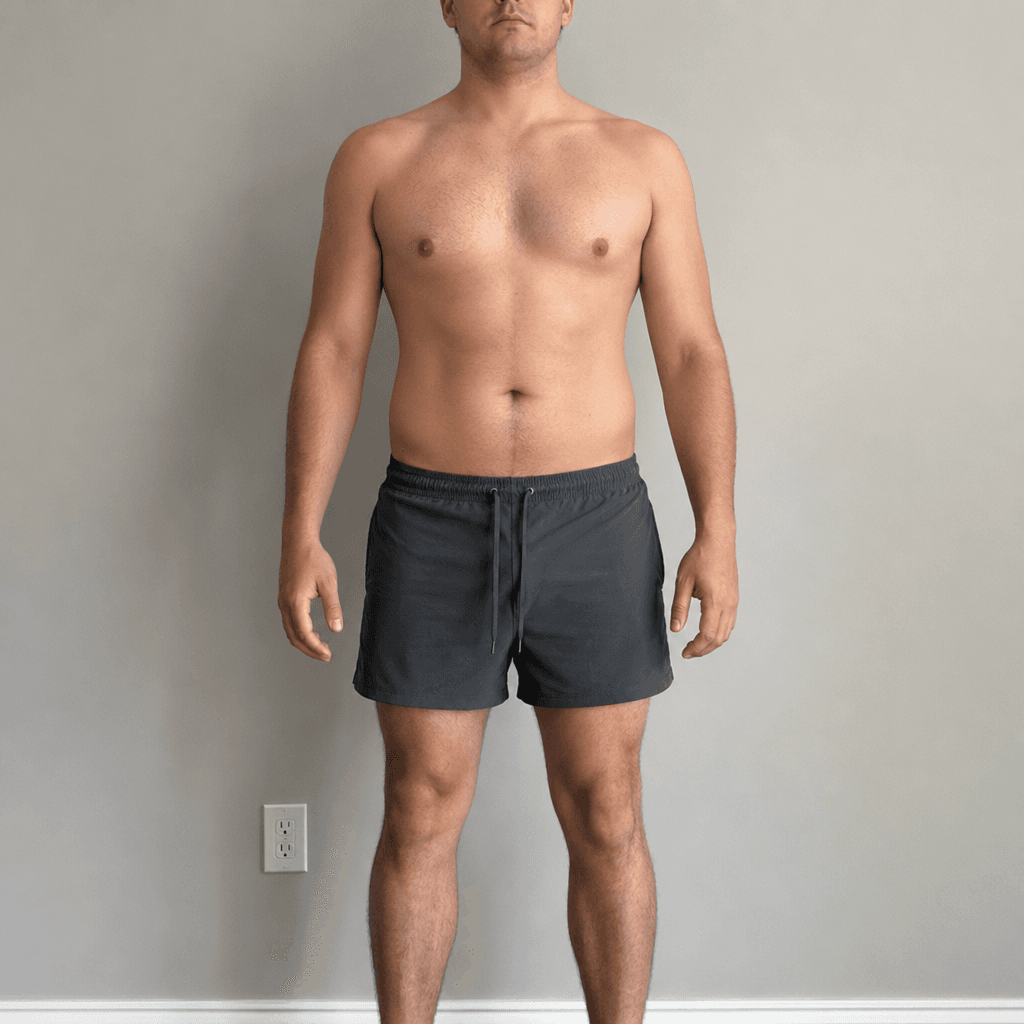
Lean Mass Gain
0.0 lbs
0.0–0.0 lbs range
Fat Loss
3.9 lbs
2.9–4.8 lbs range
Fat Loss by Week
Risks & mistakes
Common (most users)#
- Mild GI upset — nausea, loose stool, or cramping, almost always at 400–600 mg doses or when administered on a completely empty stomach. Mitigation: administer with a small fat source (fish oil cap, tbsp olive oil, or a light meal 15–20 min after dosing) and keep single doses ≤300 mg. Split 400–600 mg protocols across 2–3 pre-meal doses rather than a single bolus.
- Appetite overshoot / undereating — the compound works, and on aggressive cutting protocols users can drift below their protein floor. Mitigation: set a hard daily protein target (≥1 g/lb lean mass) and a calorie floor, and time the pre-meal dose so the satiety window aligns with an actual meal rather than skipping one.
- Mild headache or wakefulness — consistent with the PPAR-α-driven elevation of cortical acetylcholine and 5-HT seen in rodent microdialysis work. Mitigation: avoid late-evening administration if sleep onset is affected; most users dose pre-lunch or pre-dinner without issue.
- "Underwhelming" effect from bulk powder — not a side effect per se, but the most common complaint. Unmicronized raw OEA dissolves poorly. Mitigation: administer with dietary fat, or preferentially use encapsulated / micronized product.
"Daily supplementation with 125 mg OEA led to improvements in glycemic indices, oxidative stress biomarkers, and serum inflammatory factors after 8 weeks with no adverse events reported." — Darvishi et al., J Ovarian Research (2024)
Uncommon (dose-dependent or individual)#
- Persistent GI symptoms at 500–600 mg/day — if cramping or loose stool does not resolve after a week of dose splitting and food pairing, the protocol calls for a step-down to 300 mg/day.
- Shifted lipid panel — PPAR-α agonism typically improves triglycerides and hepatic enzymes (the direction seen in the Tutunchi NAFLD trial), but individual responders can see unexpected LDL movement. A 12-week lipid panel is reasonable on extended protocols.
- Subjective "flat" mood or anhedonia — rare and anecdotal; plausibly linked to appetite-reward circuit dampening. Back off to 125–200 mg/day if hedonic tone feels blunted beyond the intended food-noise suppression.
"Supplementation with OEA significantly decreased body weight, BMI, waist circumference, FBS, HOMA-IR, ALT, AST, GGT, TG, hs-CRP, TNF-α, and MDA... compared to placebo." — Tutunchi et al., Pharmacological Research (2020)
Rare but serious#
- Gallbladder symptoms — PPAR-α agonism can alter bile composition. Users with a history of symptomatic cholelithiasis who develop right-upper-quadrant pain, post-prandial colic, or referred shoulder pain should discontinue and image.
- Idiosyncratic hepatic reaction — no hepatotoxicity signal exists in the trial literature (the direction of effect on ALT/AST/GGT is favorable), but any unexplained jaundice, dark urine, or RUQ pain on an extended protocol warrants stopping and a liver panel.
- Allergic / hypersensitivity reaction to excipients — uncommon; more likely tied to the capsule formulation than to OEA itself.
Hard contraindications#
- Pregnancy and lactation — no human safety data. Do not run.
- Symptomatic gallstone disease — PPAR-α-driven changes in bile composition make this a mechanistically plausible aggravator.
- Concurrent fibrate therapy (fenofibrate, gemfibrozil) — redundant PPAR-α agonism with no documented additive benefit and unknown combined tolerability.
- Concurrent FAAH inhibitors (e.g. URB597 and related research chemicals) — blocking the primary hydrolysis pathway will produce unpredictable, massively elevated OEA exposure that no clinical dataset covers.
Sex-specific and PCT considerations#
OEA is non-hormonal and has no effect on the HPTA, androgen receptor, estrogen receptor, or SHBG. The published RCT dose (125 mg/day × 8 weeks) has been run in female PCOS cohorts with no sex-specific dose adjustment and no virilization or menstrual-cycle concerns (Darvishi 2024). No PCT is required, there is no taper, and the compound can be cycled or run continuously alongside AAS, SARMs, GLP-1 agonists, or a hair stack without interaction at any hormonal axis. Pregnancy and lactation remain the one absolute line — the rest of the profile is as clean as anything in the metabolic-adjunct shelf.
Stack & combine
Multipliers applied when these compounds run together. Values > 1 indicate a bonus on that axis. Tap a partner to expand the mechanism.
| Partner | Type | Lean | Fat loss | Recovery |
|---|---|---|---|---|
| synergistic | ×1.00 | ×1.18 | ×1.00 |
FAQ — Oleoylethanolamide
Where to buy

Swiss Chems
Affiliate link — we may earn a commission at no cost to you.
- Buy Oleoylethanolamide (OEA) 98%+ - Powder, 5 grams - SwissChems - Buy Best Quality Peptides, SARMS OnlineBuy Oleoylethanolamide
Research & citations
5 studies cited on this page.
Conclusion
Oleoylethanolamide (OEA) is a low-side-effect, research-friendly metabolic adjunct used for appetite suppression, improved fat oxidation, and liver health support, especially during cut phases. Its best-documented role is as a pre-meal satiety tool, with a simple and flexible protocol.
Key takeaways:
- Standard daily dose in human trials: 125 mg oral, taken 30–60 min before the largest meal (Tutunchi 2020; Darvishi 2024)
- Community protocols push 200–600 mg/day, often split across pre-meal doses for stronger appetite control
- Non-hormonal, non-stimulant, rapid-acting — safe for both men and women
- Cycle duration: 8–16 weeks (typical), no PCT or tapering required
- Stacks well with GLP-1 agonists (semaglutide, tirzepatide), caffeine, yohimbine, TUDCA, and berberine depending on the goal
- Best results are seen when combined with a hypocaloric diet and administered with a small dietary fat source to aid absorption
- Minimal side effect profile; GI upset at high doses is the main risk
For research targeting appetite, fat loss, or on-cycle liver support, OEA stands out as a reliable, practical, and clean addition to the metabolic stack.