Adipotide
FTPP · Fat-Targeted Proapoptotic Peptide · Prohibitin-TP01 · CKGGRAKDC-GG-D(KLAKLAK)₂
Last updated
At a glance
Overview
Why Adipotide Earns Its Niche#
Adipotide (FTPP) is the most mechanistically elegant fat-loss peptide in the underground market. Unlike GLP-1s, which work through appetite and gastric emptying, or AOD-9604, which nudges lipolysis, adipotide does something genuinely novel: it prunes the microvasculature feeding white adipose tissue, triggering ischemic involution of the fat depot itself. The homing peptide CKGGRAKDC docks onto a prohibitin–annexin A2 receptor complex enriched on WAT capillaries; the D-(KLAKLAK)₂ domain then collapses the mitochondrial membrane of the targeted endothelial cell. Fat tissue loses its blood supply and regresses.
The data that put it on the map came from rhesus primates at MD Anderson, where a 28-day course produced double-digit body-weight loss and a near-halving of insulin requirement — with no effect in lean controls.
"On average, obese monkeys lost 11 percent of their body weight and 39 percent of their total fat mass... Weight loss was accompanied by near‐doubling in insulin sensitivity." — ScienceDaily summary of Barnhart et al., 2011
For physique-focused users, the specific appeal is visceral and stubborn-depot adiposity — the residual fat that remains after GLP-1s plateau, the visceral accumulation that builds up on heavy GH/slin protocols, the insulin-resistant metabolic profile that diet alone won't unwind. Adipotide is second- or third-line behind diet, GLP-1s, and GH-axis peptides, but it occupies a slot nothing else can fill: a short, finite, non-hormonal course that selectively targets WAT vasculature and leaves lean mass untouched.
The trade-off is a real renal signal. The kidney clears the peptide and concentrates it in proximal tubule cells, which is why community protocols run at or below the primate dose rather than above it, and why aggressive bloodwork is non-negotiable. The sections below cover the documented dose ladder from the Barnhart primate data, subcutaneous protocol design, stack placement alongside GLP-1s and GH fragments, the renal monitoring cadence that keeps the compound viable, and the side-effect profile and hard contraindications that decide who should and shouldn't run it.
How Adipotide works
Adipotide is a chimeric peptidomimetic — two functional domains stitched together by a Gly-Gly linker. The first domain (CKGGRAKDC) is a homing motif that recognises a receptor complex found almost exclusively on the capillaries feeding white adipose tissue. The second domain, D-(KLAKLAK)₂, is a pro-apoptotic warhead that destroys mitochondria once it gets inside a cell. The mechanism is therefore ligand-directed vascular pruning: adipotide doesn't mobilise fat, suppress appetite, or accelerate lipolysis. It kills the blood supply feeding the fat depot, and the adipocytes involute for lack of perfusion.
Vascular Homing via the Prohibitin–ANXA2 Complex#
Adipotide was identified through phage-display vascular mapping, which flagged prohibitin (PHB) as differentially expressed on WAT endothelium relative to endothelium elsewhere in the body. The CKGGRAKDC homing sequence binds a surface PHB–annexin A2 (ANXA2) heterocomplex enriched on the capillaries irrigating white fat. This is what gives the molecule its tissue selectivity — the warhead alone would be indiscriminately cytotoxic, but the homing domain concentrates delivery at adipose vasculature.
"A synthetic peptide targeting adipose vasculature induced selective apoptosis of endothelial cells, leading to rapid depletion of white adipose tissue in obese mice, without affecting other tissue types." — Kolonin MG et al., Nature Medicine, 2004
Practical consequence: activity scales with available target. Lean subjects have less WAT vasculature to prune and therefore respond less — a feature that caps the compound's utility in already-lean physique users and explains the flat response reported in leaner community logs.
Mitochondrial Disruption by D-(KLAKLAK)₂#
Once bound and internalised, the D-amino-acid (KLAKLAK)₂ segment — an amphipathic α-helix resistant to proteolysis thanks to its D-stereochemistry — inserts into the inner mitochondrial membrane, collapses the membrane potential, and triggers caspase-dependent apoptosis of the targeted endothelial cell. Capillaries die, the downstream adipocyte depot loses perfusion, and the fat mass is reabsorbed over the following weeks.
This is why the fat loss lags the dosing window: body-weight curves in the primate data kept declining for roughly three weeks after the last injection, as ischemic involution played out.
Secondary Effect on Fatty-Acid Uptake#
The PHB–ANXA2 complex isn't just a docking site — it also scaffolds CD36-mediated fatty-acid transport into adipocytes. Disrupting the complex reduces lipid influx into remaining depots, adding a second, non-apoptotic contribution to the overall fat-loss signal.
"These results show that the prohibitin–ANXA2 complex regulates fatty acid uptake in adipose tissue and functions as the vascular target for adipotide." — Salameh A et al., JCI Insight, 2016
This likely explains why insulin sensitivity improves before the bulk of the mass loss has occurred: reduced ectopic lipid loading and WAT inflammation precede the final body-composition change.
Metabolic Reprogramming of Residual WAT#
Beyond raw mass loss, the primate data show that the remaining adipose tissue behaves differently post-cycle — lower inflammatory tone, reduced serum triglycerides and free fatty acids, and markedly improved whole-body insulin action.
"Adipotide caused up to an 11% reduction in body weight and significant improvements in insulin resistance, while lean monkeys exhibited no change in weight." — Barnhart KF et al., Science Translational Medicine, 2011
In the same rhesus cohort, obese subjects lost roughly 39% of total fat mass and doubled insulin sensitivity across a 28-day protocol — a metabolic shift that cannot be explained by mass loss alone and points to genuine reprogramming of residual WAT.
Adiposity-Dependent, Not Depot-Selective#
A persistent community misconception is that adipotide performs "spot reduction" at stubborn depots. The imaging data don't support this. The MRI and DEXA readouts from the primate work showed proportionate loss across visceral and subcutaneous compartments, scaling with the local density of PHB-positive vasculature rather than with any specific anatomic site. Users chasing a particular depot will see reduction there only in proportion to its share of total WAT — which, for visceral fat, is actually favourable, since visceral depots are densely vascularised.
The Renal Tradeoff Is Mechanistic, Not Incidental#
The kidney is the clearance organ, and the proximal tubule concentrates the peptide during reabsorption. This is why the dose-limiting toxicity is renal, not hepatic or cardiac — it's a direct consequence of how the molecule is eliminated, not an off-target surprise. Hydration, avoidance of concurrent nephrotoxins, and mid-cycle renal bloodwork are therefore not optional add-ons; they are built into the mechanism itself.
Protocol
| Level | Dose | Frequency | Notes |
|---|---|---|---|
| Low | 0.25–0.3 mg/kg | Once daily | Documented entry-level range |
| Mid | 0.3–0.4 mg/kg | Once daily | Most commonly studied range |
| High | 0.4–0.5 mg/kg | Once daily | Daily SC dosing, calculated against lean body mass in community practice. Injection sites rotated across the abdominal fat pad. |
Cycle length & outcomes
Documented cycle
2–4 weeks
Plateau after
7 wks
Cycle Structure & Protocol Length#
Adipotide is not a compound that rewards extended exposure. The renal signal is cumulative, and the Barnhart primate data — the only serious efficacy dataset in existence — capped at 28 days for a reason. Documented community protocols run shorter and lower than the published primate dose, with a mandatory washout between any repeat exposure.
The compound works via ligand-directed vascular pruning of white adipose tissue. Each daily dose drives a small additional increment of endothelial apoptosis in WAT capillary beds; the effect accumulates over days-to-weeks rather than pulsing acutely. Weight continues to decline for 2–3 weeks after the last dose in the primate model, which means the cycle length on paper underreports the total effect window.
Dose Ladder by Goal#
| Goal | Cycle Length | Daily Dose (SC) | Frequency |
|---|---|---|---|
| First exposure / conservative recomp | 14 days | 0.25 mg/kg | Once daily |
| Visceral fat cut (intermediate) | 21 days | 0.30–0.40 mg/kg | Once daily |
| Post-GLP-1 plateau break | 21 days | 0.30 mg/kg | Once daily |
| Aggressive recomp (advanced, Barnhart analog) | 28 days | 0.40–0.50 mg/kg | Once daily |
Dose is calculated against lean body mass in community practice, not total body weight — the peptide's activity is proportional to available WAT vascular target, and lean-mass scaling modestly reduces the delivered dose in higher-BF subjects where the kidney load matters most.
Loading, Tapering, and Onset#
There is no loading phase. A single day's dose does not meaningfully accelerate onset; the mechanism is cumulative endothelial apoptosis, and front-loading only front-loads the renal exposure.
There is also no taper. The peptide has no receptor-mediated downregulation to ease off and no endogenous axis to restore. Dosing stops on the scheduled last day.
Onset timing from the Barnhart primate data:
- Days 1–5: no visible change. Subclinical endothelial apoptosis is underway.
- Days 5–10: reduced food intake begins (a documented effect, not a flaw — welcome during a cut).
- Days 10–14: measurable body-weight decline begins; insulin sensitivity improvements precede most of the mass loss.
- Days 21–28: peak on-cycle weight loss.
- Days 28–49: continued post-cycle decline as the adipocyte involution completes. This is the plateau point for body-composition re-measurement.
Bloodwork Cadence#
This is the non-negotiable part of the protocol. The renal signal is the dose-limiting toxicity, and it's the reason the community dose sits at-or-below the published primate dose rather than above it.
| Timepoint | Panel |
|---|---|
| Baseline (≤7 days pre-cycle) | CMP, urinalysis with microalbumin, cystatin C, eGFR |
| Day 10–14 (mid-cycle) | CMP, urinalysis with microalbumin |
| Day 30 (post-cycle) | CMP, urinalysis, cystatin C, eGFR |
| Day 90 (pre-repeat gating) | Full renal panel — must return to baseline before any future cycle |
Cystatin C is preferred over creatinine alone for mid-cycle monitoring because it catches early tubular dysfunction before creatinine moves. Rising creatinine, new proteinuria, or a falling eGFR mid-cycle is a stop signal, not a "reduce dose" signal.
Body-composition measurement (DEXA or quality bioimpedance) is taken at day 0 and again at day 49 — measuring earlier underreports the result because the post-cycle decline is still in progress.
"Adipotide caused up to an 11% reduction in body weight and significant improvements in insulin resistance, while lean monkeys exhibited no change in weight." — Barnhart et al., Science Translational Medicine (2011)
That lean-subject null is the other structural constraint on cycle design: the peptide's activity scales with available WAT vascular target, so lean physique-focused users have less to prune and should not chase the result by pushing dose or duration. The right response to a weak signal is to stop the cycle, not extend it.
Repeat Cycles & Washout#
One cycle is the default. Serial cycling is uncommon and discouraged.
- Minimum washout: 90 days between cycles.
- Gating requirement: full renal-panel normalization, confirmed at the 90-day post-cycle draw.
- Maximum annual exposure (community consensus): two cycles per year, and only when the first cycle's renal markers returned cleanly to baseline.
"A synthetic peptide targeting adipose vasculature induced selective apoptosis of endothelial cells, leading to rapid depletion of white adipose tissue in obese mice, without affecting other tissue types." — Kolonin et al., Nature Medicine (2004)
The short, one-off cycle structure is what makes adipotide defensible despite the renal profile. Run it tight, run it with bloodwork, and run it rarely — the mechanism does the work on a timeline that doesn't reward heroics.
Body Transformation Preview
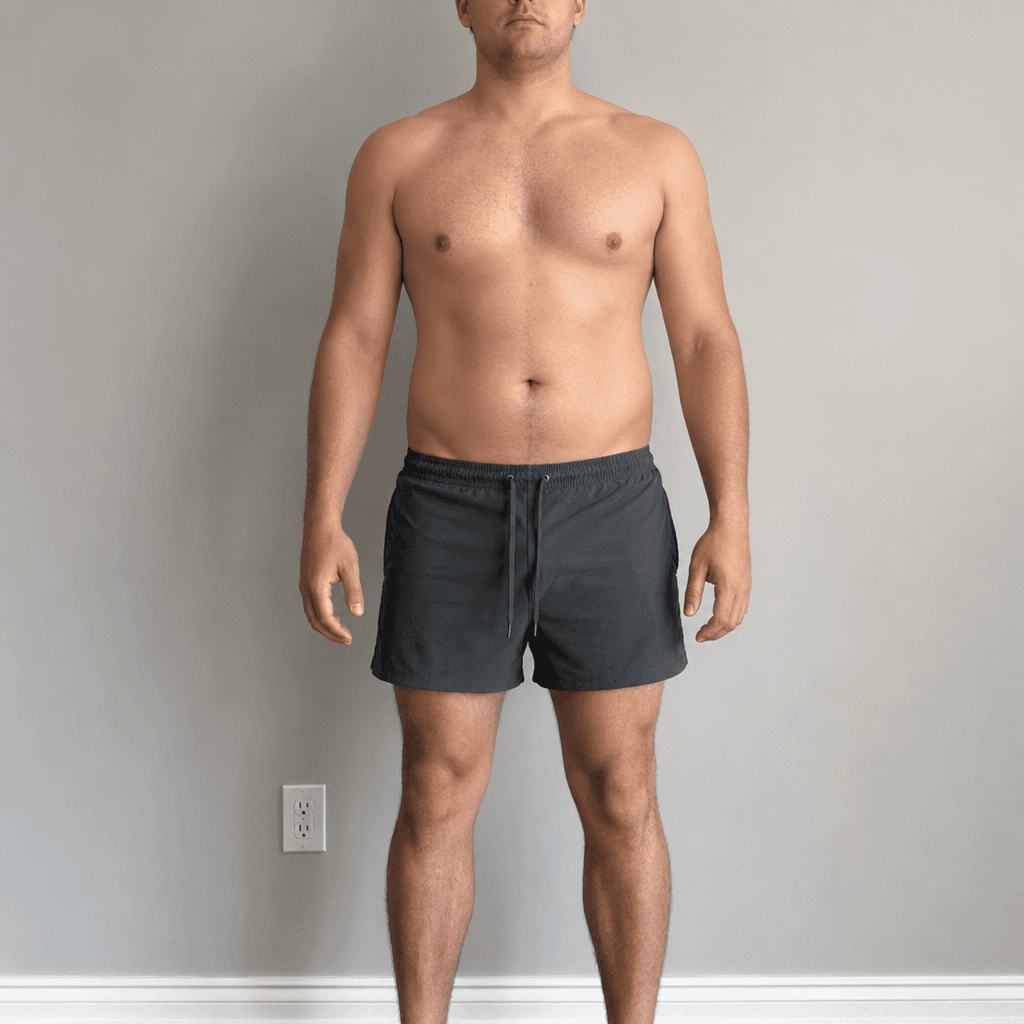
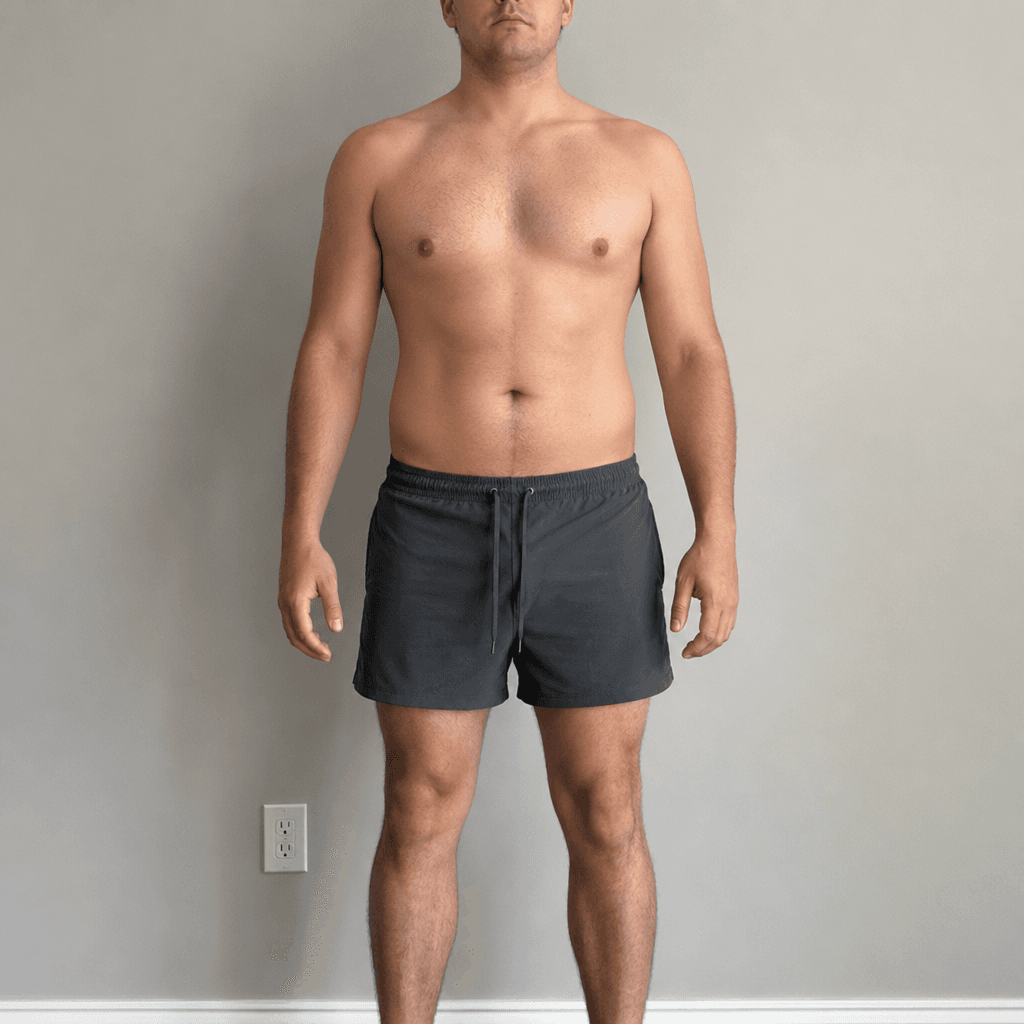
Lean Mass Gain
0.0 lbs
0.0–0.0 lbs range
Fat Loss
3.0 lbs
2.2–3.7 lbs range
Fat Loss by Week
Risks & mistakes
Common (most users)#
- Injection-site reactions — mild erythema, bruising, or tenderness at the SC site. Rotate across quadrants of the abdominal fat pad; avoid reusing a site within 72 hours. Ice the site post-injection if tender.
- Transient appetite reduction — onset typically day 5–10, welcome for a cut but worth tracking. Maintain protein intake (≥1g/lb lean mass) and electrolytes even when caloric intake drops.
- Mild thirst / increased urination — expected given renal clearance. Baseline hydration of 3–4 L/day is non-negotiable; add electrolytes (sodium, potassium, magnesium) rather than plain water alone.
- Low-grade fatigue in week 1 — usually resolves as the body adapts. If it persists past day 10, it's a prompt to check a mid-cycle renal panel rather than push through.
Uncommon (dose-dependent or individual)#
- Rising serum creatinine / falling eGFR — the signature dose-limiting signal. Any rise above baseline at the mid-cycle draw is reason to hold the dose; a 20%+ rise is reason to stop. Cystatin C picks this up earlier than creatinine and should be on the panel.
- Microalbuminuria / new proteinuria — earliest tubular-stress marker and the reason urinalysis is part of the baseline and mid-cycle workup. New proteinuria ends the cycle.
- Electrolyte shifts — sodium and potassium disturbances secondary to tubular dysfunction. Caught on the same CMP that tracks creatinine.
- Menstrual irregularity — reported anecdotally in female subjects, mechanistically consistent with prohibitin expression on ovarian vasculature. Grounds to discontinue and defer future cycles.
- Additive appetite suppression when stacked on GLP-1s — not harmful in itself but can drive unintentional under-eating. Track intake; maintain protein floor.
"Adipotide caused up to an 11% reduction in body weight and significant improvements in insulin resistance, while lean monkeys exhibited no change in weight." — Barnhart et al., Science Translational Medicine (2011)
The lean-subject null result is the other side of the efficacy story: leaner users have less WAT vascular target, so less systemic exposure of the homing peptide to productive binding sites — and proportionally more to spill into renal clearance. Leaner physique-focused users should start at the low end (0.25 mg/kg) and treat the renal signal as the ceiling, not the dose.
Rare but serious#
- Acute kidney injury with incomplete recovery — the Barnhart primate data described renal effects as dose-dependent and reversible, but those were 28-day observations. Independent reports in the community describe cases where renal markers did not fully normalize at the 90-day post-cycle check. Warning signs: creatinine rise >30% from baseline, new significant proteinuria, oliguria, or flank pain. Stop immediately; do not attempt another cycle.
- Dehydration-driven prerenal injury — the interaction between cutting-phase caloric and fluid restriction and a renally-cleared peptide is the most preventable serious adverse event. A cutting diet without electrolyte management is not the context to run adipotide.
- Off-target vascular effects — prohibitin is expressed beyond WAT endothelium (bone marrow, ovarian, and some tumor vasculature). No clinical case series documents this in humans, but any unexplained cytopenia, reproductive symptom, or new mass during or after a cycle warrants workup rather than a wait-and-see.
"A synthetic peptide targeting adipose vasculature induced selective apoptosis of endothelial cells, leading to rapid depletion of white adipose tissue in obese mice, without affecting other tissue types." — Kolonin et al., Nature Medicine (2004)
The selectivity holds in animal models at documented doses. It is not a license to freelance on dose or duration.
Hard contraindications#
- Pre-existing renal impairment — any baseline eGFR <90, known CKD, single kidney, or history of nephropathy. The kidney is both the clearance organ and the dose-limiting toxicity target; there is no safe adipotide protocol for a compromised renal baseline.
- Baseline proteinuria — closes the door on adipotide entirely.
- Concurrent nephrotoxic agents — NSAIDs (including daily low-dose), aminoglycosides, cisplatin-class chemotherapeutics, high-dose IV contrast within the cycle window. Swap NSAIDs to acetaminophen for the duration.
- Active malignancy — prohibitin expression on some tumor vasculature makes the effect unpredictable; not a compound to run while a cancer workup or treatment is active.
- Pregnancy or attempted conception — the peptide induces endothelial apoptosis; there is zero reproductive-safety data.
- Dehydration states — aggressive water-cuts, contest prep peak-week protocols, endurance events in heat. Run adipotide in a well-hydrated metabolic phase or not at all.
- Concurrent heavy oral AAS cycle — the combined renal and hepatic load is not worth it. Schedule adipotide into a non-cycle window or a bridge.
Gender-specific considerations and PCT#
Adipotide has no interaction with the HPG axis, no androgen-receptor activity, no aromatase effect. PCT is not required. No hormonal ancillaries apply.
Female subjects face one additional consideration: prohibitin expression on ovarian vasculature raises a theoretical concern for follicular or menstrual disruption, and anecdotal community reports echo this. Female users with active fertility plans should defer entirely. Female users without fertility plans should still treat any menstrual change during or after a cycle as a signal to discontinue and defer future exposure until a full workup clears it.
The final framing: adipotide's side-effect profile is dominated by one organ system. Get the renal baseline right, hydrate aggressively, check the mid-cycle panel, and stop on any signal — and the compound behaves. Skip the monitoring and it is one of the more dangerous peptides on the research market.
FAQ — Adipotide
Where to buy

Swiss Chems
Affiliate link — we may earn a commission at no cost to you.
- Buy FTPP (Adipotide) 5 mg (1 vial) - SwissChems - Buy Best Quality Peptides, SARMS OnlineBuy Adipotide
Research & citations
5 studies cited on this page.
Conclusion
Adipotide is the most mechanistically unique fat-loss peptide in the research arsenal — with primate data showing real visceral-fat targeting, but a renal signal that demands respect and proper monitoring.
Key takeaways:
- Community protocol: 0.25–0.5 mg/kg (lean mass) SC once daily, 14–28 days max
- Route: subcutaneous, rotating abdominal injection sites; reconstitute carefully and refrigerate
- Washout: minimum 90 days between cycles; serial cycling is discouraged
- Monitoring: baseline and mid-cycle CMP, urinalysis (microalbumin), cystatin C; strict hydration (3–4 L/day)
- Stacking: most effective following a GLP-1 plateau; avoid concurrent NSAIDs, orals, or other nephrotoxins
- Feature benefit: can drop 0.6–0.8 lb/week of fat in the right subject, with insulin sensitivity improvements documented in primate studies
For research on aggressively diet- and GLP-1-resistant visceral fat, adipotide earns its slot — assuming renal markers stay immaculate and cycle design stays within documented practice.
"On average, obese monkeys lost 11 percent of their body weight and 39 percent of their total fat mass... Weight loss was accompanied by near-doubling in insulin sensitivity."
— Barnhart et al., Science Translational Medicine (2011); ScienceDaily summary